I’m trying to do the embryo culture after I read (over and over) the Donald Holeman’s text. I’ve trying to extract embryos from moist and estratified seeds… FAIL… I did squish the little babies, They was too soft… So I tryied to extract embryos from seeds imediately harvested… FAIL… The pericarp was too attached, the babies were broken. I won’t give up, but I need some advice from someone more experienced about How can I prepare seeds to embryo extraction? May I dry the seeds before? How long?
Rodrigo,
I haven’t tried it. Here’s an article by someone who has.
Karl
Hi Karl,
This text is my guide already. I’m thinking the seed parent have influence about It… I finally did some embryo extractions sucessfully with seeds from Glacier Magic, and that was very more easy. I put my first embryos under a LED lamp… If It works I’ll put my offspring in potted soil in 10-15 days from the harvest instead several months. Thank you Karl for always help us, new breeders.
Sincerely,
Rodrigo
Keep us posted on your results, Rodrigo.
I have been tentatively planning to try some embryo extraction this year, because I have some crosses that I feel are important but have been getting low germination in recent years. I plan to practice on OP seed from the same female parent before attempting to do the crosses. If even 50% of my attempts are successful it would be an improvement over my conventional sowing of the seeds.
The real question is if my personality is suited to embryo extraction. The temptation to cut some corners of Don’s rigorous methods is strong.
I believe Don says that it is just fine to dry the seeds thoroughly and store them before extraction, although they will lose vitality as time passes. Just from memory, not sure.
Hi Jbergeson,
Ok, I’ll show my results in few days (goods or not).
I lose a lot of embryos, as you I began with OP seeds, three hips later I started to have sucess when I take the risk of using one of my crosses. In that hip I did have 50% rate of sucess (in big seeds - I didn’t try with the smaller ones). I wasn’t so religious with Donald’s methods. I just focused in keep all things clean using Donald’s peroxyde solutions, the nail clips really worked well to open seeds, and you will need something like a needle to remove the testae. When you try to do, sounds more easy than when you reading or even explaining to someone. Learn the principles and you will find your own way. Good luck.
Rodrigo
If the seeds have been removed from the hips immediately prior to embryo extraction…and the hips themselves are fresh, sanitary precautions as outlined in Don Holeman’s article are much less important. This will simplify the process for you as well as improve your overall germination success. As well, using a proper magnifying device is crucial. If I’m damaging more embryo’s than usual, I take a break from the process…give my eyes and arms a rest…put on some classical musical…and start up again. This has been my experience.
Joe…it takes a special personality to extract embryos from a single rugosa hip…40-50 seeds per hip…torture…I save them for the last.
I know, I know… It’s early, but is very exciting to see the embryos becoming green just three days after extracting.
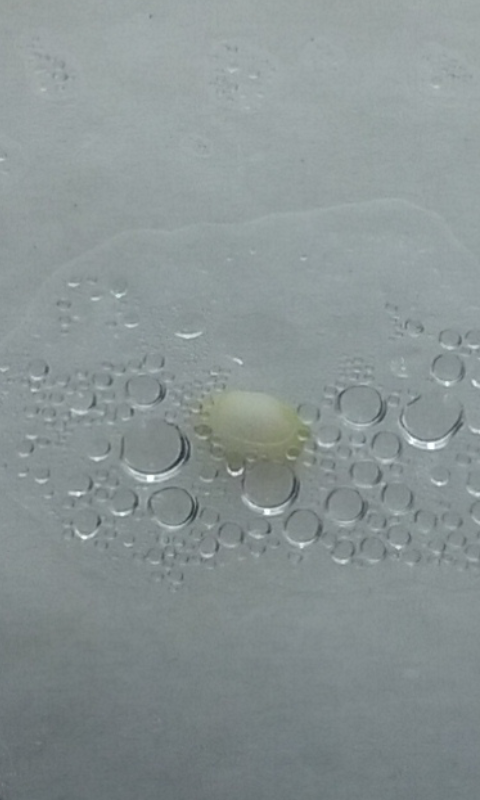
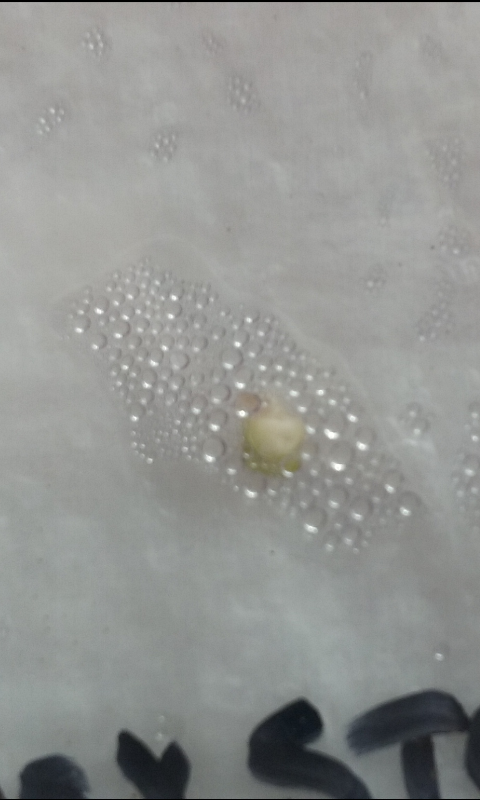




With 5 days now.

Rodrigo,
I am happy for you. I hope you post more pictures as your embryos grow.
I also thought it would be worth mentioning that many plants have been grown from embryos taken from immature seeds. One advantage is that there is no hard shell to remove.
http://bulbnrose.x10.mx/Heredity/Tukey/Tukey_Growth/Tukey_Growth.html
Karl
![]()
Fresh seeds are more difficult to extract embryos from than dried ones but not impossible. The key is to apply forces tangentially to the surface of the seed using a very sharp tool. Principles that apply to cutting things with knives apply here as well - the finer the finish and the more acute the angle the more successful you will be.
Brand new nail clippers are never optimally sharpened during manufacture. Moreover, there is a lot of variability in such details as the angle of sharpening, the fineness of the finish, the spacing of the jaws and so forth. If you have a rotary tool such as a Dremel it is easy to sharpen and resharpen them yourself using very fine paper disk abrasive.
My toolkit has grown to now include many clippers in many sizes from many different brands because, over time, even the best known brands are inconsistent in quality and dimension.
If the seeds have been removed from the hips immediately prior to embryo extraction…and the hips themselves are fresh, sanitary precautions as outlined in Don Holeman’s article are much less important.
Hmm. It is true that you can successfully germinate extracted embryos without dogmatic adherence to a sanitary protocol.
Embryos die and disintegrate from many causes during incubation. Often times the cause is not infection or contamination but very often it is. If you succeed without sanitary precautions it is because the infectious agent is not present within or on the embryo or because the embryo is vigorous enough to resist the disease.
I think your observation that fresh hips and proximate extraction mitigate the need for sanitary protocols is correct but only to the extent that your breeding stock is healthy and vigorous, and that winds from afar don’t bear ill spores at extraction time. I can’t prove it but waiting for cold, clean Canadian air to move in seems to help as much as any other voodoo.
Rodrigo, I am interested in seeing more updates…those things look so tiny and delicate in the last pic. It seems like getting them to the real leaf stage should be difficult.
I just finished collecting all of my hips and I have tons this year but some seemingly important crosses where I might recover remontancy in a small percentage of seedlings and want to have as many chances as possible so I am planning in addition to typing very long run-on sentences to try my hand at embryo extraction for the first time but I’m a little scared about my abilities.
Joe, any virginianas in those croses?
Don,
Hell yeah. I had my first blossoms on [First Impression x R. virginiana] #3 this spring and I used every pollen grain, figuratively. It proved to be very pollen fertile to the degree that if I spotted a particularly fat labeled hip I knew before looking that the label said “FIVI3”.
When crossing this rose with a modern tetraploid or triploid rebloomer I would expect 1/6 or fewer of the seedlings to be remontant. Yet it would be so nice to have a remontant rose that is 25% R. virginiana. So every seed is precious.
(some seeds using FIVI3 as a seed parent, but it showed a strong tendency to abort hips)
I also had blooms on two plants of All A’Twitter x R. virginiana. So I used that quite a bit both ways. However, Rob Byrnes said his specimen of AAT x VIRG broke down with blackspot pretty badly, so I’ve not as much enthusiasm for these crosses. It would still be fun to get a reblooming offspring.
The first generation virginiana crosses show so much hybrid vigor. I’m very curious to see if their offspring will share that vigor. Especially if a seedling showed great vigor and was also remontant, although those two characteristics seem averse to showing up in the same plant together.
I had poor germination last year. Sometimes a whole flat full of 400+ seeds would yield only a handful of germinations. That’s why I’m going to try to extract this winter.
Sometimes a whole flat full of 400+ seeds would yield only a handful of germinations. That’s why I’m going to try to extract this winter.
I forget the exact numbers but somewhere here I posted my actual tallies for seeds/embryos/germinations. IIRC net germinations are in the 5% if-you-are-lucky range. With your space availability you could realize greater efficiency by going for big numbers of seeds and sticking to chants and dances for germination.
Really? Five percent if lucky even when extracting?
Gosh, I’m really tempted to just sow them all. I have the will to put in some hours on my rose breeding, but I am definitely a lazy person. And I have enormous quantities of seeds and crosses to deal with.
I am now considering doing a warm and cold stratification in baggies with Perlite as some people do, then doing the actual sowing in early March. I wonder how much of a pain it would be removing the seeds from the perlite. Maybe I could fold them up in a little envelope of dry paper towel before placing in the bag with moistened perlite.
5%, really, after all is said and dead. There is a very wide variability though. If somebody could find that post it also gives the number of seeds and number of embryos.
I don’t want to misquote myself incorrectly but the number of embryos as a percent of number of seeds runs very low too. The single biggest reason that rose seeds do not germinate is that they have no embryos. Dead embryos and embryos with intrinsic disease spores are the second and third biggest reasons seeds do not germinate.
My advice would be to practice extracting with lower value seeds until you gain enough confidence to extract your highest value seeds; stratify everything in between.
Several years ago Jim Sproul posted here that his preferred method of stratification eliminates all media, just soak the the seeds, drip dry a bit and bag them in the fridge. I second that. Cold water throughout the process is critical, too. Warming wet seeds suffocates them - the solubility of oxygen in water decreases with increasing temperature - and it gives disease spores an opportunity to hatch. Diplocarpon requires 50 degrees F or better to germinate, for instance (ULB Bonn :: Dissertationen + Habilitationen online).
45 - 48 degrees F is the optimal temperature range for germinating stratified seeds (in my personal experience). Keep them ice cold but not frozen throughout stratification, bring them to 45 - 48 degrees until germinations starts then stops, rinse and repeat stratification.
I like your description of Jim’s method. Wouldn’t hurt to have some ZeroTol and calcium nitrate in the soak, eh? And thanks for the tip about the cold water. So they should be put in the fridge for the duration of the soak, I hear you saying. Which should be no more than overnight or 24H, I assume.
I was going to just put the seeds back into the bags in which I had the hips from that cross. Wonder about disease spores in the bags. Can’t seem to justify buying and writing on hundreds of new bags, tho. Maybe a quick ZeroTol rinse if visual debris in bag.
I tried going at my very first two seeds with a fingernail clipper today and quickly realized I might not have the patience for extraction.

Uptading pictures of my seedlings. Three seedlings are thriving, in the middle is (Sangerhauser jubilaumsrose x Sterntaler) and the others are (SJ x Bianca).They’re slow growers and I didn’t have seen a bloom yet, They never got diseases, until now. My seedlings are full of powdery mildew.


Did you know some commercial or homemade solution?
Thanks,
Rodrigo
I’ve used 1 part milk mixed with 5 parts water and sprayed every few days on full grown rose bushes for powdery mildew and it’s worked. However I’ve never tried on seedlings.